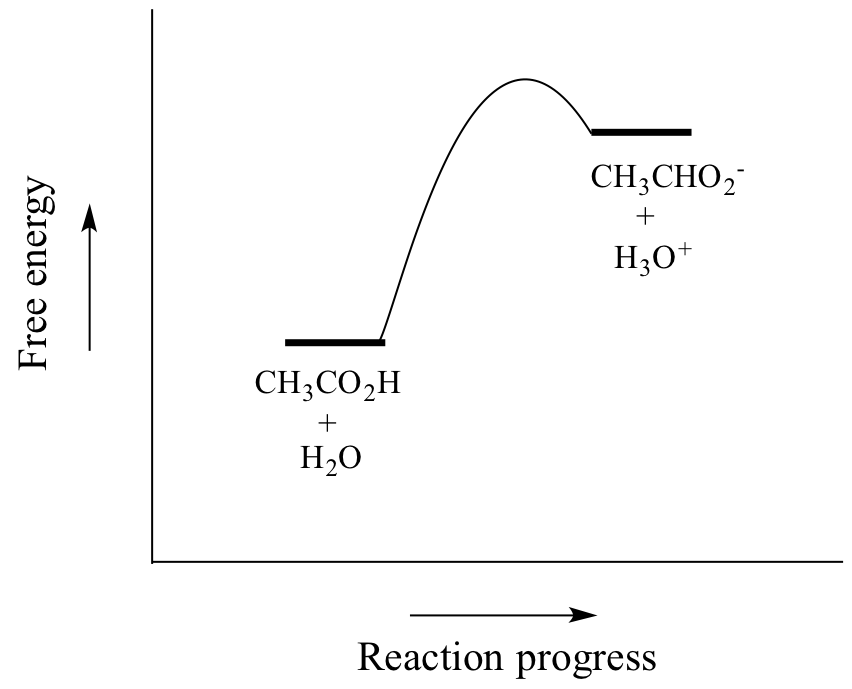
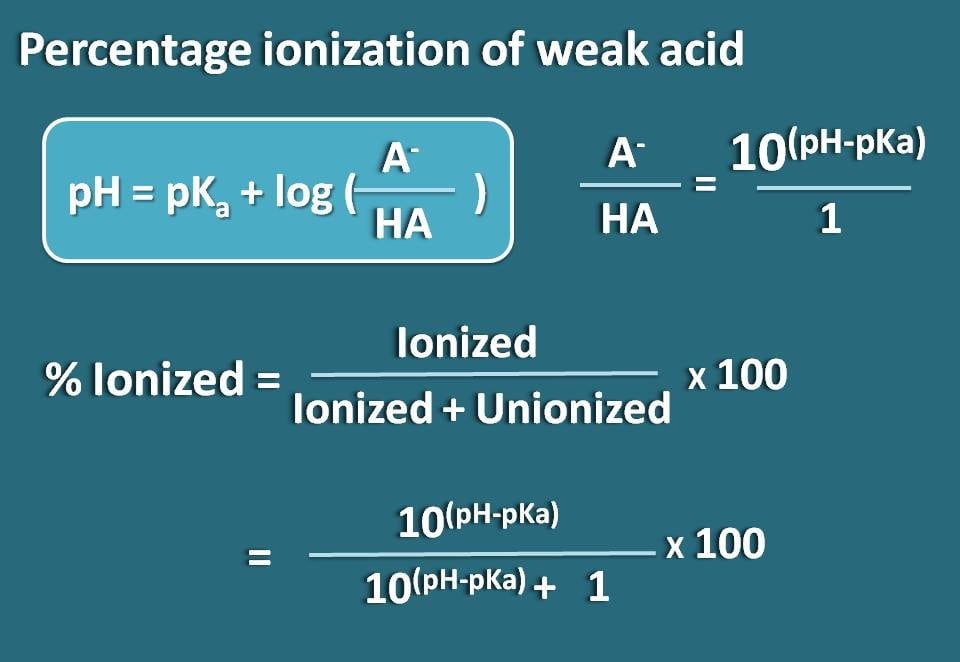
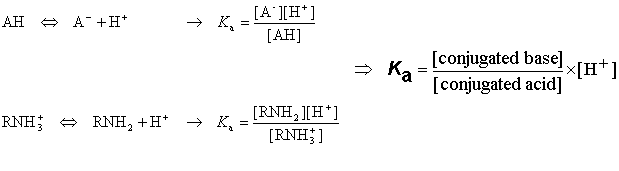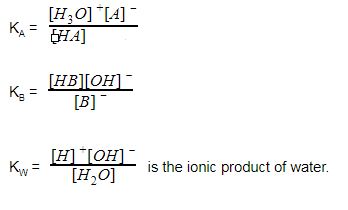How to Calculate Analyte Concentration Using the Equivalence Point in an Acid-base Titration | Chemistry | Study.com

Calculate the the ratio of conjugate base// weak acid required to prepare an aqueous solution of... - YouTube

Ionization of Acid And Bases - Arrhenius concept of Acid And Base Ionisation, Explanation, Determination ionisation constant of Acid base, Examples And FAQS

Determining the acid and conjugate base concentrations using the Henderson-Hasselbalch equation - YouTube
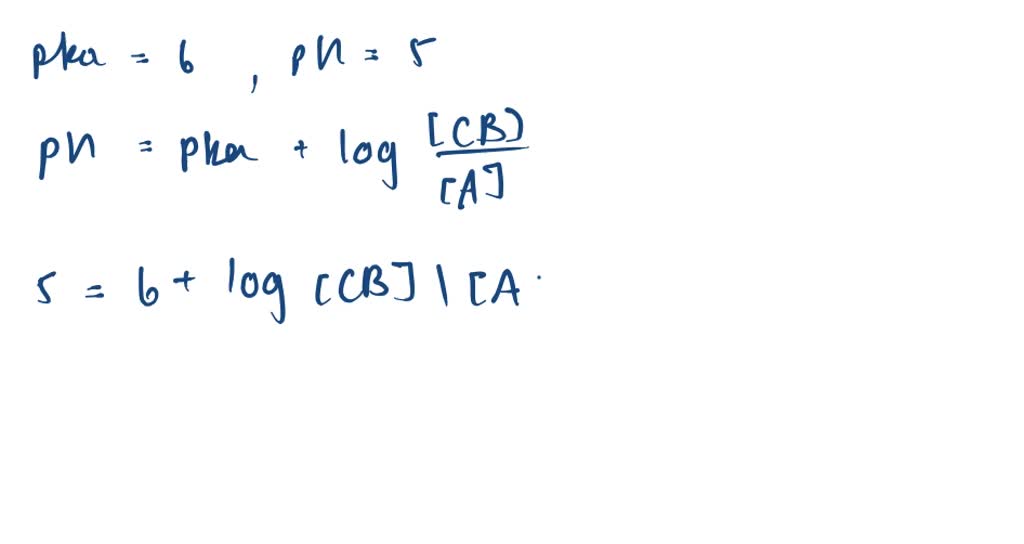
SOLVED:Calculation of Molar Ratios of Conjugate Base to Weak Acid from pH For a weak acid with a p Ka of 6.0, calculate the ratio of conjugate base to acid at a
![SOLVED: Acid/Base Ratio Unanswered 1 attempt left What ratio of acid to base is needed to prepare a buffer with a pH = 3.5 using the conjugate pair HCOOH/HCOO 1(Ka 1.78x10-4J? [HCOOH]/[HcOO-] = SOLVED: Acid/Base Ratio Unanswered 1 attempt left What ratio of acid to base is needed to prepare a buffer with a pH = 3.5 using the conjugate pair HCOOH/HCOO 1(Ka 1.78x10-4J? [HCOOH]/[HcOO-] =](https://cdn.numerade.com/ask_images/3e3ccf6738d048629ccc88b39c08e146.jpg)