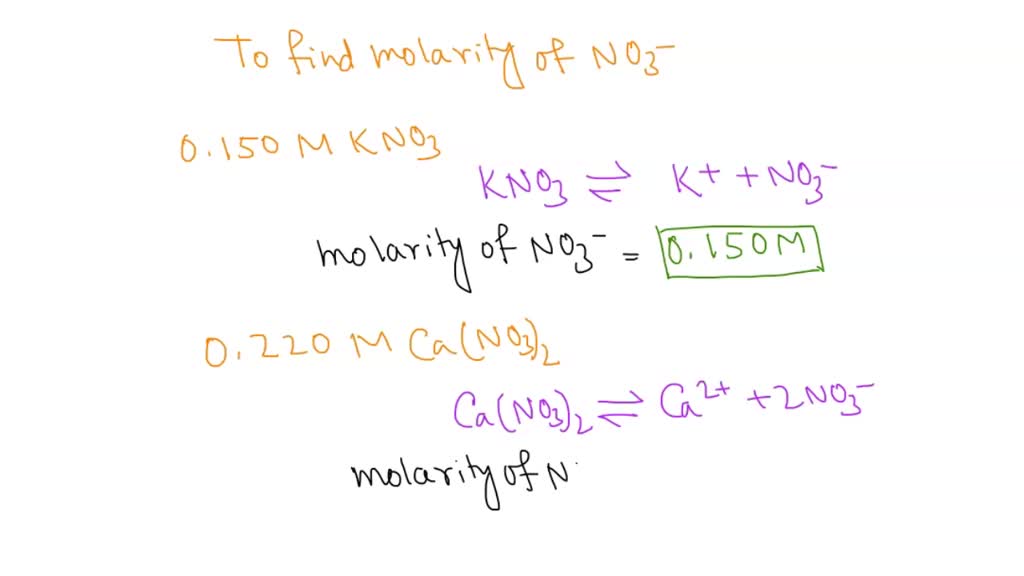
SOLVED: What is the molarity of NO?3 in each solution? 0.150 M KNO3. 0.220 M Ca(NO3)2 0.370 M Al(NO3)3.

Molarity vs. Molality: Examples | How to Calculate Molarity & Molality - Video & Lesson Transcript | Study.com

Calculate the molarity of a solution that is 39.77% H2SO4 by mass. The specific gravity of the solution is 1.305. | Homework.Study.com

What will be the molarity of the solution in which 0.365 g of HCl gas is dissolved in 100 mL of solution?

Dilution Equation & Examples | How to Calculate Dilution Factors - Video & Lesson Transcript | Study.com

SOLVED: What is the molarity of Cl- in each solution? a. 0.200 M NaCl b. 0.150 M SrCl2 c. 0.100 M AlCl3

Calculate the molarity of NaOH in the solution prepared by dissolving its 4 g in enough water to... - YouTube

Molarity, Molality, Volume & Mass Percent, Mole Fraction & Density - Solution Concentration Problems - YouTube

Converting molarity to mole fraction, mass percent and molality: A 0.750 M solution of H(2)SO(4) in water has a density of 1.049 g mL^(-1) at 20 ^(@)C. What is the concentration of














