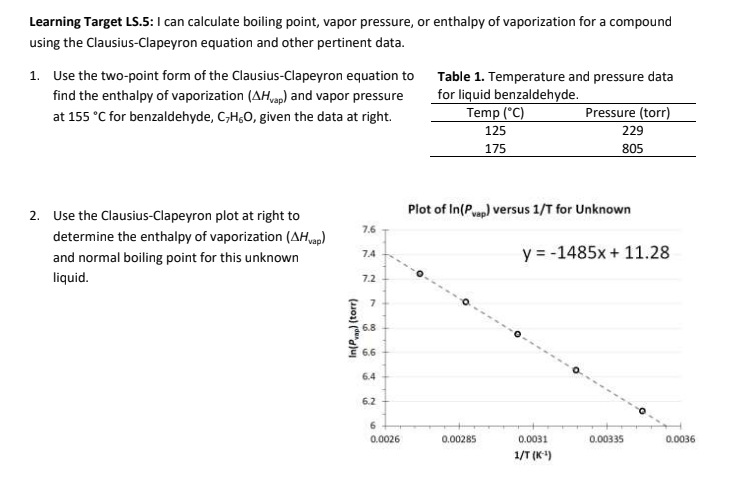
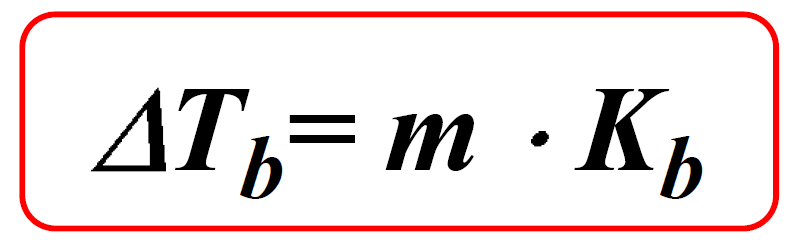SOLVED: Question 4 1pts The vapor pressure of a liquid is 300 torr at 51.0 *C,and its enthalpy of vaporization is 37.66 kJlmol. Calculate the normal boiling point of this liquid in *

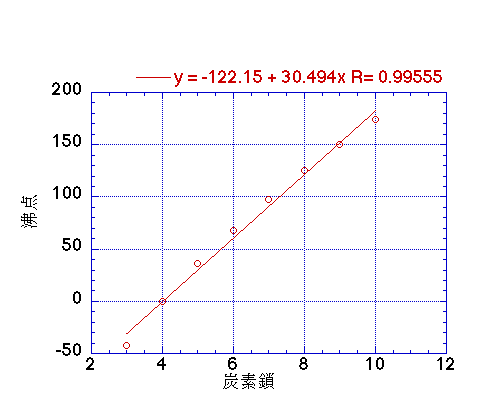
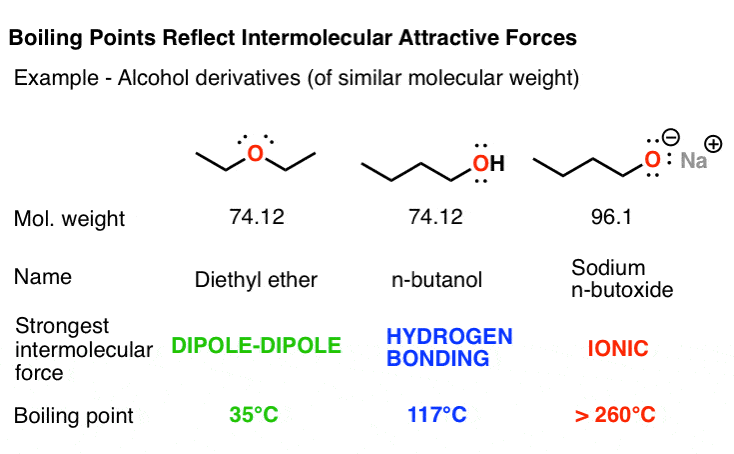
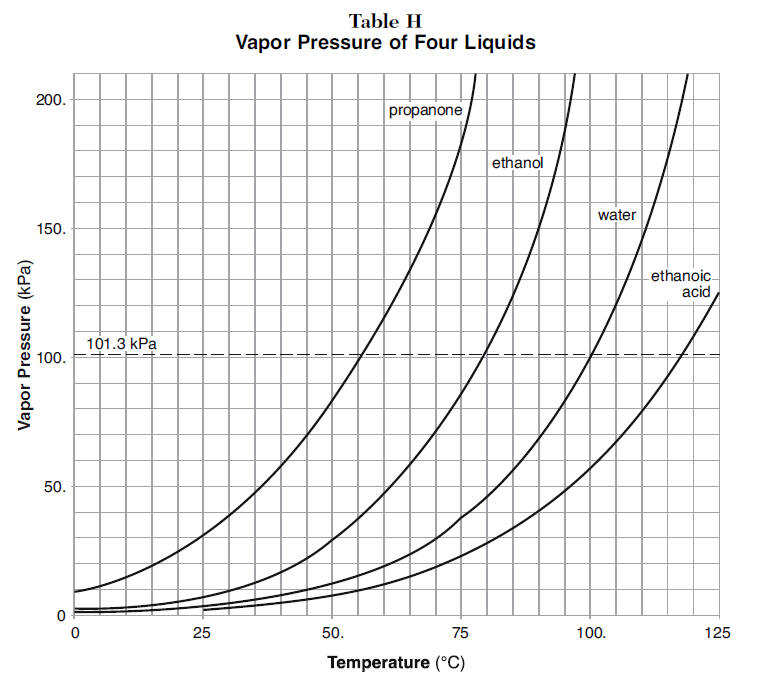
intermolecular forces - How can I determine the highest boiling point given a list of molecules? - Chemistry Stack Exchange
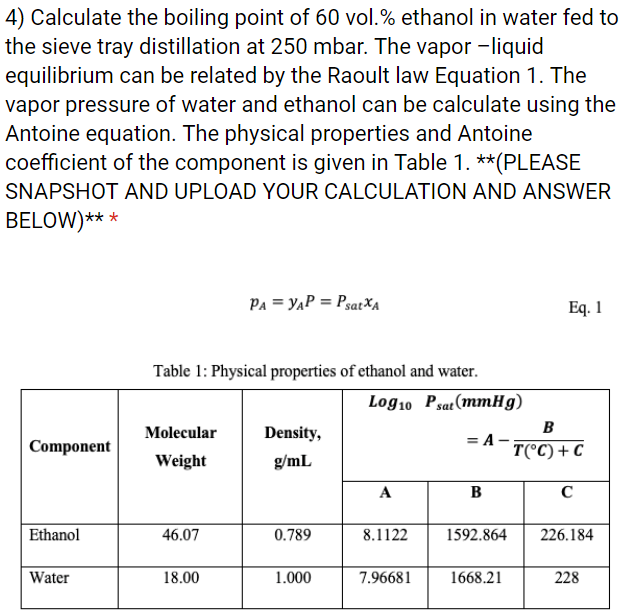
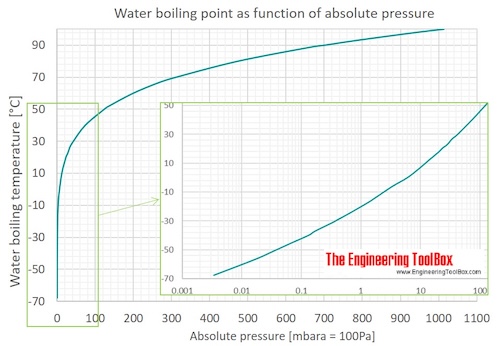
How does one calculate the boiling point of water at different pressures without a steam table? - Quora

Boiling Point Elevation and Freezing Point depression - Example 2 ( Video ) | Chemistry | CK-12 Foundation