Phenolic Aldehyde Epoxy Vinyl Ester Resin for Higher Heat and Corrosion Resisting Required Product - China Resin and Unsaturated Polyester Resin

Acetalization of poly(vinyl alcohol) by a fatty aldehyde in water medium: Model study, kinetics, and structure analysis - Piluso - 2018 - Journal of Polymer Science Part A: Polymer Chemistry - Wiley Online Library
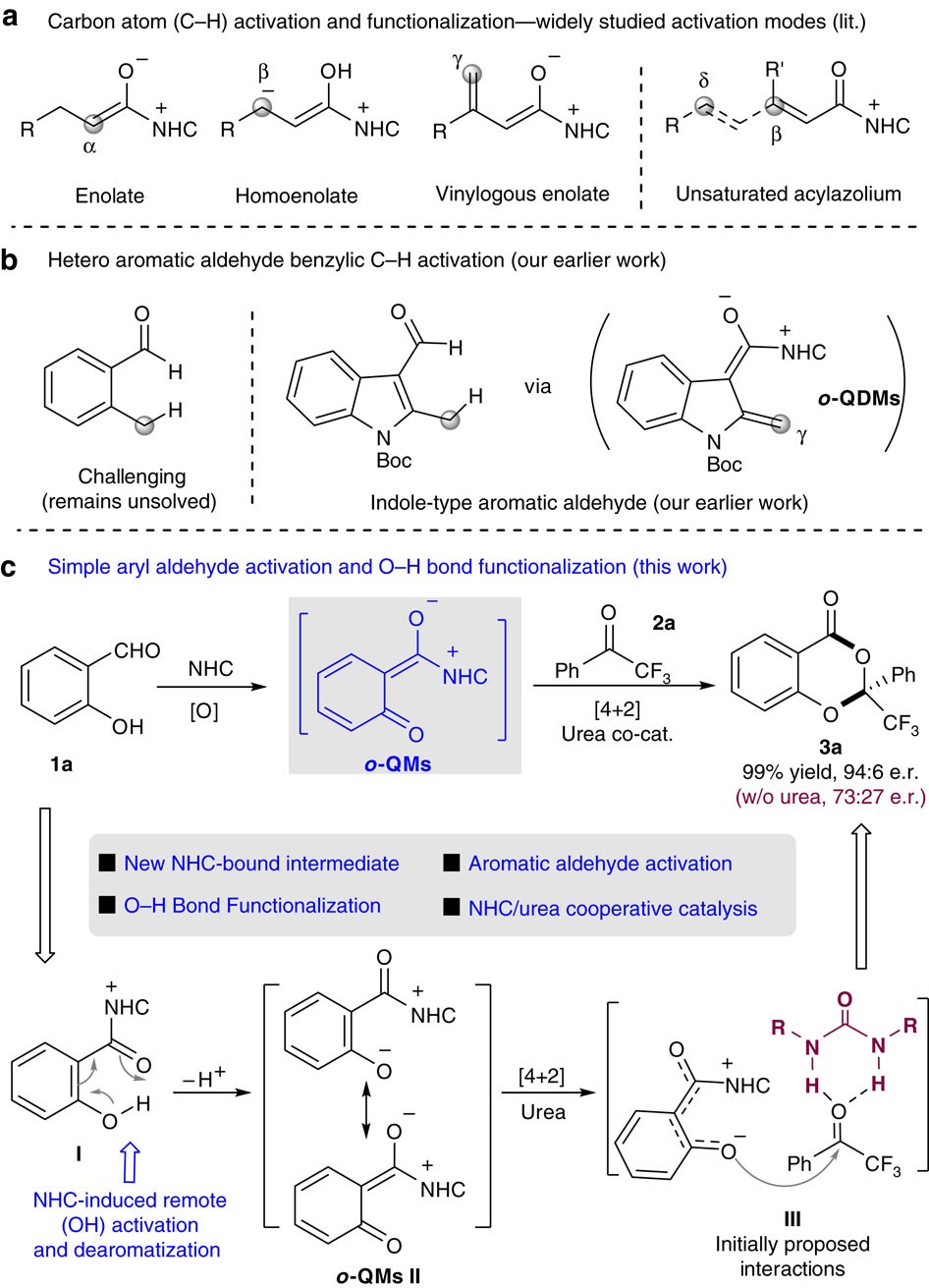
A reaction mode of carbene-catalysed aryl aldehyde activation and induced phenol OH functionalization | Nature Communications

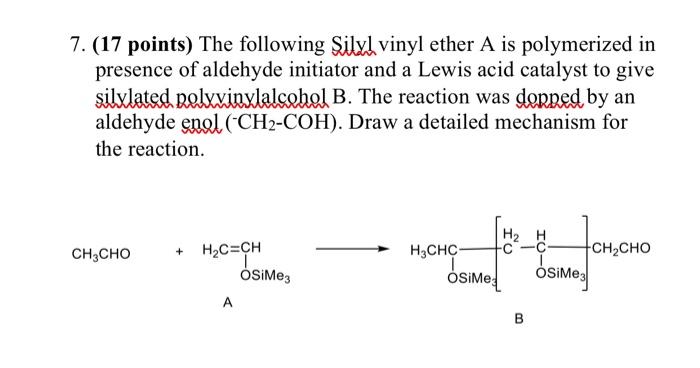
As CH2=CH is called vinyl group Can we call it vinyl aldehyde - Chemistry - Organic Chemistry Some Basic Principles and Techniques - 10963770 | Meritnation.com

Pentafluorophenyl vinyl sulfonate enables efficient, metal-free, radical-based alkene hydroacylation with an aldehyde as a limiting reagent - RSC Advances (RSC Publishing) DOI:10.1039/C5RA08353B
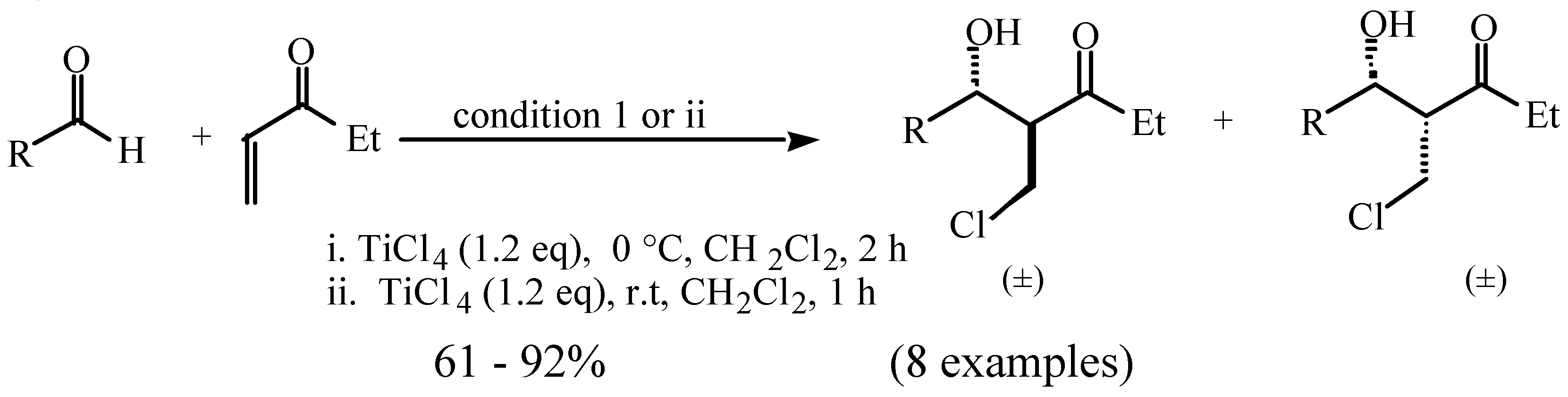
Molecules | Free Full-Text | Halogeno Aldol Reaction of Ethyl Vinyl Ketone and Aldehydes Mediated by Titanium Tetrachloride
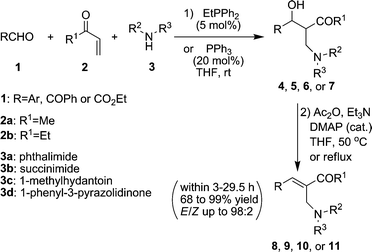
Organocatalytic tandem three-component reaction of aldehyde , alkyl vinyl ketone , and amide : one-pot syntheses of highly functional alkenes - Organic & Biomolecular Chemistry (RSC Publishing) DOI:10.1039/C0OB00644K

Asymmetric Vinylogous Aldol-type Reactions of Aldehydes with Allyl Phosphonate and Sulfone - ScienceDirect

Vinyl chloride-induced interaction of nonalcoholic and toxicant-associated steatohepatitis: Protection by the ALDH2 activator Alda-1 - ScienceDirect
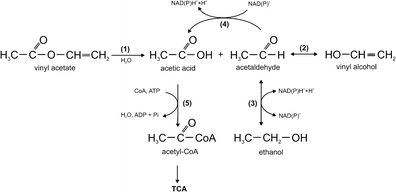
Enzymes involved in vinyl acetate decomposition by Pseudomonas fluorescens PCM 2123 strain | SpringerLink

Enantioselective α-Vinylation of Aldehydes via the Synergistic Combination of Copper and Amine Catalysis | Journal of the American Chemical Society
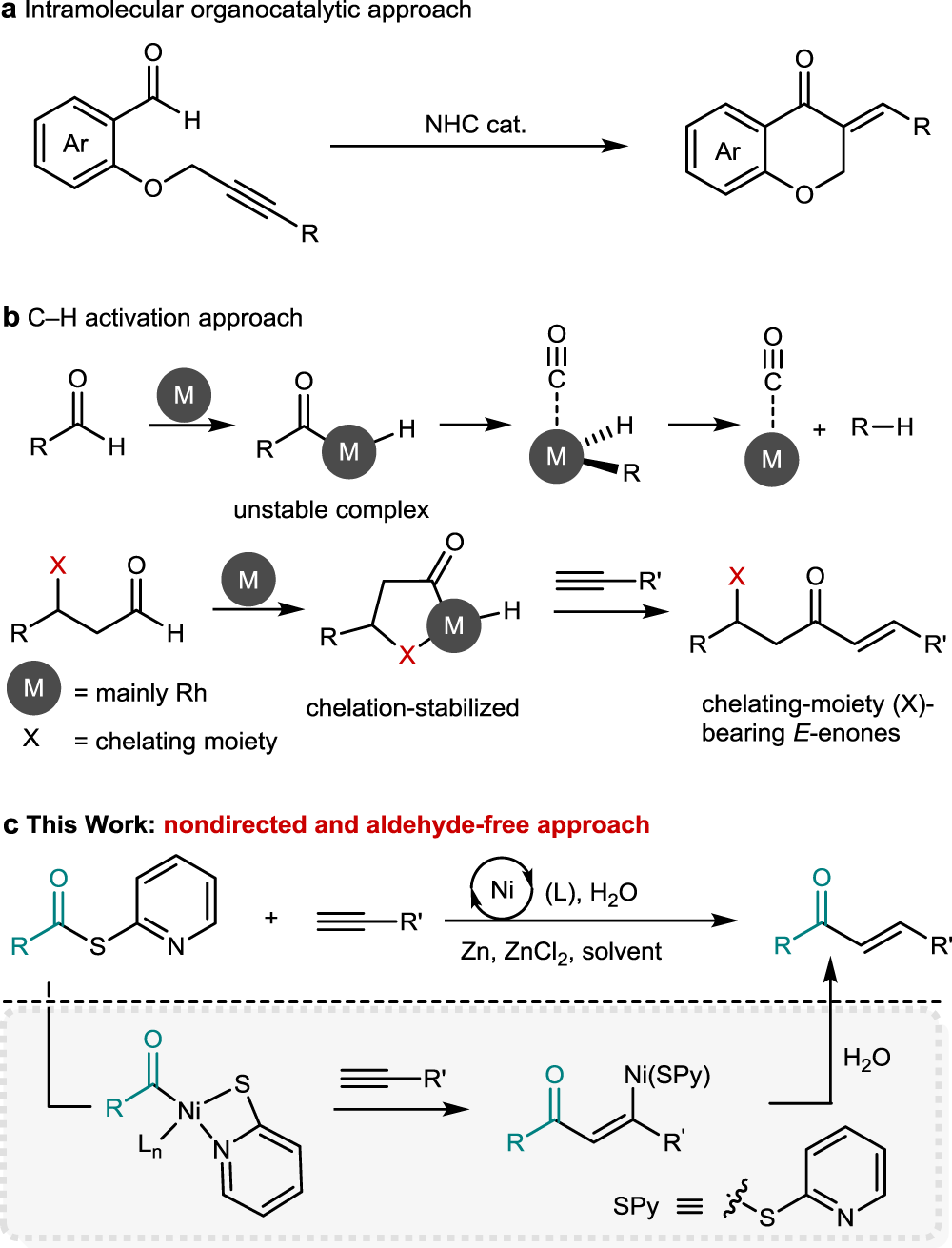
Synthesis of α,β-unsaturated ketones through nickel-catalysed aldehyde-free hydroacylation of alkynes | Communications Chemistry
Scheme 56. Synthesis of vinyl azides and azirines starting from the... | Download Scientific Diagram

Synthesis of Functionalized α‐Vinyl Aldehydes from Enaminones - Chen - 2019 - Angewandte Chemie International Edition - Wiley Online Library

Enantioselective α-vinylation of aldehydes via the synergistic combination of copper and amine catalysis. - Abstract - Europe PMC

Traditional Morita-Baylis-Hillman reaction of aldehydes with methyl vinyl ketone co-catalyzed by triphenylphosphine and nitrophenol