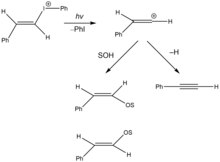
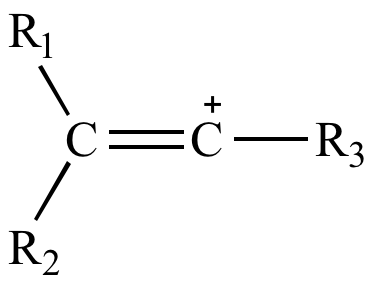
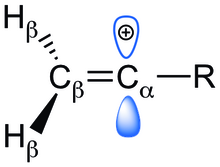
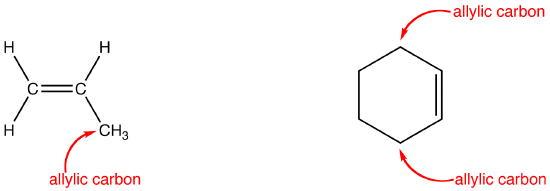
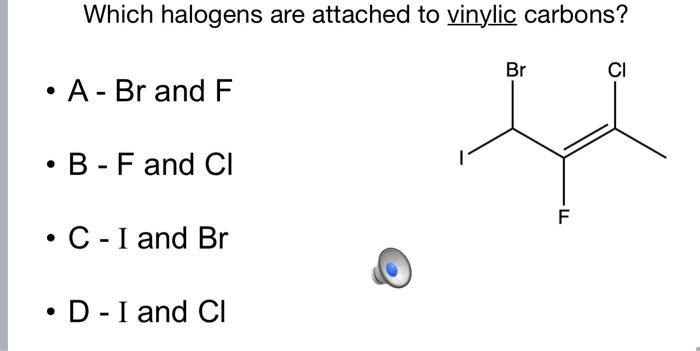
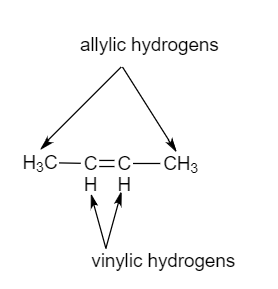
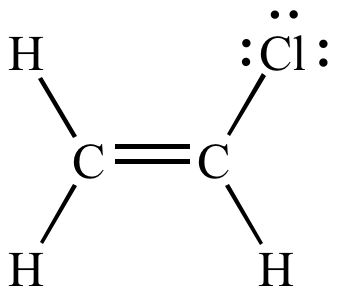
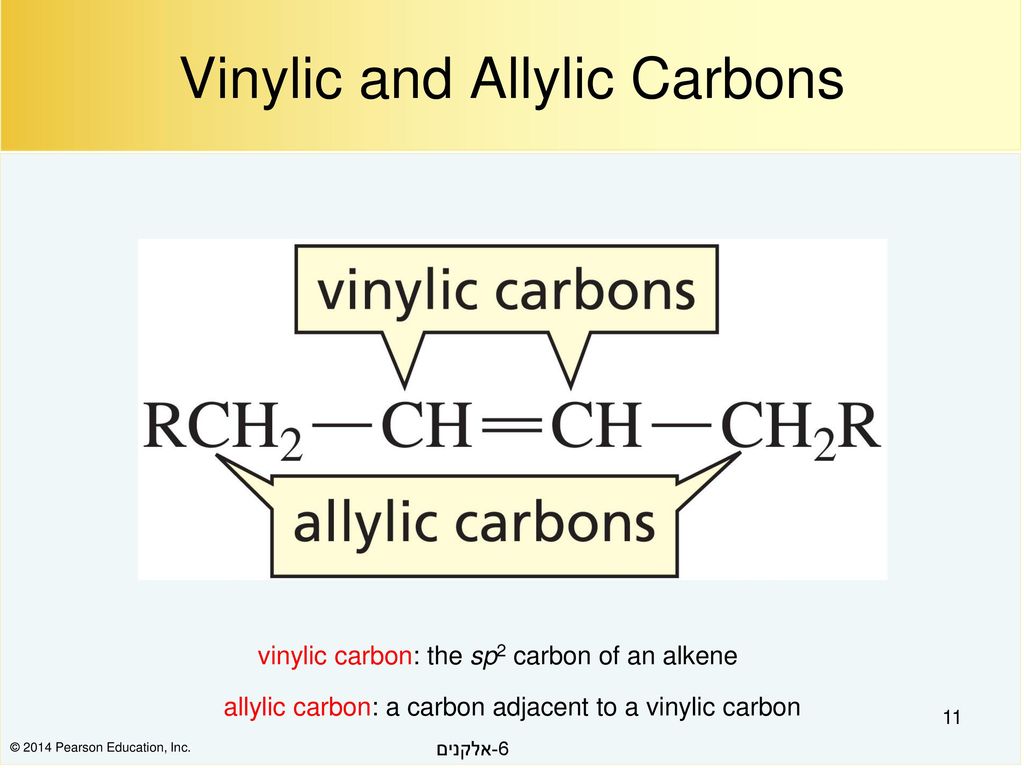
Rare Example of Nucleophilic Substitution at Vinylic Carbon with Inversion: Mechanism of Methyleneaziridine Formation by Sodium Amide Induced Ring Closure Revisited | Journal of the American Chemical Society

Bimolecular nucleophilic substitutions at the saturated sp 3 and vinyl... | Download Scientific Diagram
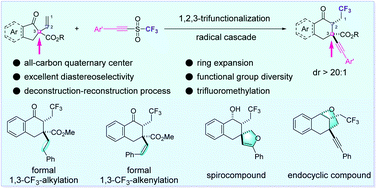
Radical 1,2,3-tricarbofunctionalization of α-vinyl-β-ketoesters enabled by a carbon shift from an all-carbon quaternary center - Chemical Science (RSC Publishing)

Long-range coupling between protons more than two carbon atoms apart is sometimes observed when pi bonds intervene. An example is found in 1-methoxy-1-buten-3-yne. Not only does the acetylenic proton, Ha, couple with